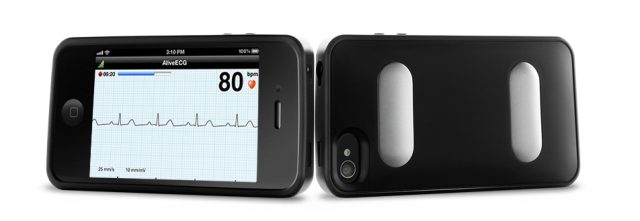
The next time your doctor says, “I have an app for that,” try not to punch them in their stethoscope. Why? Because they just might be using AliveCor. For those unfamiliar, the San Francisco-based company is the maker of a low-cost, clinical-grade mobile heart monitor (fondly known as an electrocardiogram, or ECG, monitor) that fits over the back of your iPhone.
This is not the first we’ve heard of nifty mobile ECGs or heart monitors. (See our coverage of Mio, Cardiio and SmartHeart, for example.) AliveCor has been in development since 2008, but today it became part of an important step forward for mobile health (and heart health), as it received FDA approval — or 510(k) clearance — for its mobile heart monitor.
FDA approval is not something that comes easily, or happens quickly, and for that reason many startups avoid constructing (or pitching) their apps, services or software as “medical” devices, that prescribe advice or treatment and rather go for utility as a general health or wellness service. But co-founders Dr. David Albert, Bruce Satchwell and Kim Barnett said recently that their “aspirations are significant” and that they’re out “to make a difference,” and FDA clearance was the first step. (In fact, the founders were granted U.S. Patent No. 8,301,232 for the device and technology. An auspicious one-two punch for AliveCor.)
With FDA Class II clearance under its belt, AliveCor is now selling its ECG monitor on its website for $199. To receive one, doctors need to input the necessary information (and ID numbers) to prove that they’re in fact registered physicians and, within the next few months, AliveCor will begin more aggressively reaching out to doctors to encourage them to prescribe the monitor to their patients, likely at a lower price (around $99), according to MobiHealthNews. If the company receives its next order of 510(k) clearance from the FDA, it will then begin selling its device over the counter in drug stores and the like.
As to how it works: AliveCor’s ECG monitor comes with two electrodes embedded in casing that can be snapped onto the back of an iPhone 4 and 4S. The device is then launched via the startup’s corresponding iPhone apps, which allows a patient to take ECG readings by either placing the sensors directly over their chest or on their fingers.
Once the reading is taken, the ECG data is transmitted wirelessly to the company’s cloud service from the heart monitor through its proprietary communication protocol, which requires no pairing between the iPhone and the device. This allows patients to store their readings in AliveCor’s secure cloud database, where they can be accessed for later analysis, or shared and printed via its website.
In terms of use cases, the co-founders see the device as providing an easy way to screen for cardiac arrhythmias or atrial fibrillation, both in-office and remotely, allowing them to monitor at-risk patients or those who’ve recently had surgery. As someone who has undergone heart surgery (ablation as well as having been treated with medicines), I am somewhat biased, but can attest to the importance of having a device that allows one to easily and remotely monitor heart rates post-surgery — and being able to do so via a casing that is compatible with the devices we carry with us everywhere? That has real life-saving potential.
The company now wants to create a more universal casing that is compatible with the different generations of the iPhone, as well as with Android devices. In addition, according to MobiHealthNews, AliveCor also plans to launch a “pad version” of its heart monitor, which will allow patients to get ECG readings by placing the palm of their hand on the device. This could then be implemented in doctor’s offices or health kiosks. All of these will require additional FDA approval, so today’s clearance was just the first step in an ongoing process that will take time.
But it’s getting easier to imagine a time when, a year or two from now, AliveCor (or a device like it) will be compatible with all of the major mobile form factors and available over the counter in major drug store chains and in doctors’ offices. While we all get plenty of mileage (and enjoyment) from the advances mobile technology has allowed in photo-sharing, social networking and finding restaurants, what mobile tech can do for health can be truly life-changing — and life-saving.
Soon, we’ll be able to detect heart abnormalities before they reach a critical point and, using data collected from our always-in-pocket devices, advance our understanding of correlations between behavior and risk, and so on. Technology still has most of its utility or application in what happens after a medical event or trauma, but the more it moves toward prevention, the more it can make a real difference.