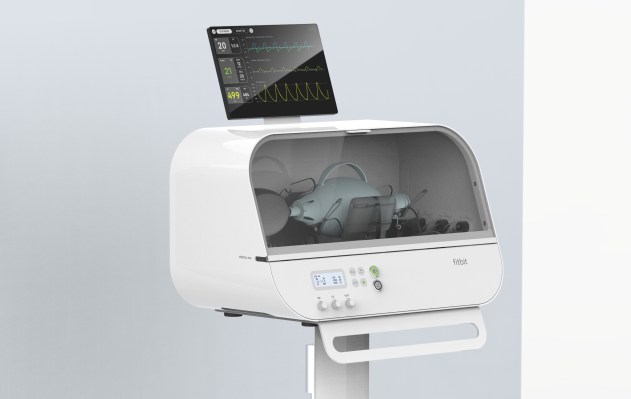
Fitbit has secured an Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) for its Fitbit Flow emergency ventilator. The ventilator hardware is low-cost, and doesn’t require very much training or expertise to use, making it a good solution for deployment in scenarios where healthcare systems are overwhelmed by resource strain stemming from the COVID-19 pandemic.
The Fitbit ventilator is based on the MIT E-Vent system, as well as specs provided by the U.K. government for ventilators to be used by hospitals in that country during the ongoing coronavirus outbreak. It’s an automated resuscitator-style ventilator, which essentially replicates the function of the types of manual resuscitation bags used by paramedics and EMTs in the field.
This is a style of emergency ventilator that has become popular in light of the pandemic, in part because they can be built using relatively affordable and readily available components versus the standard style of medical ventilators healthcare facilities typically use. Fitbit says it believes that its design is particularly effective, with the right combination of sensors, automated alarms and other patient-monitoring features that supplement the automation of resuscitation bag pump.
While a lot of the attention around the need for emergency use ventilators has subsided in recent weeks, the need still exists, and will likely resurge along with new waves of COVID-19 transmission in the coming months. Projects like the Fitbit Flow aim to provide options should they be required, and the FDA EUA means that the company can now work with its existing manufacturing partners to build these in large volumes to address need.
Ventilators like the Flow aren’t designed to replace existing, traditional medical ventilators — instead, they’re intended as stopgaps, to be used only when that hardware isn’t available in quantities needed to treat patients.