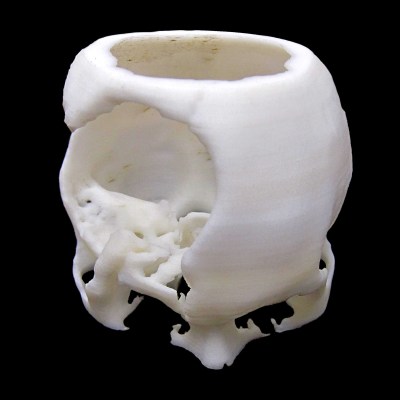
The campus of Washington State University in Southeast Washington’s agricultural region looks like a typical land-grant university. The connected mix of art-deco, modern, and post-modern buildings that collectively house the College of Engineering and Architecture hide a strange and incredible secret: that the researchers inside are close to making human-compatible ceramic bone grafts and custom-made prostheses and implants. In short, they’re building cyborgs in Palouse.
Welcome to the future of 3D-printed body hacks. Dr. Susmita Bose and Dr. Amit Bandyopadhyay have been waiting for you.
In the same way that commercial 3D printing has changed product design and prototyping, these researchers are creating a way to build body parts. Their cheap, iterative designs take the best of the 3D-printing industry and add novel materials like resorbable ceramic powders and titanium. The resulting artificial body parts can then be placed on humans, creating some of the most complex chimeras in existence.
“Using 3D-printing technology, and optimum material chemistry, one can control the geometry and shape of the scaffold and bone-like material chemistry at the same time. We can control the resorption and dissolution kinetics in a controlled manner that can be used based on application need. If you can resorp the scaffold, then the ultimate result would be for the natural healing to replace the scaffold and need for a second surgery, as is needed with current technologies,” explains Dr. Bose on a recent visit to her office.
The calcium phosphate-based ceramic bone grafts, for low-load-bearing bones, and titanium prostheses, for high-load-bearing applications, that the MRG is making are not ready for human use yet, but Dr. Bose and Dr. Bandyopadhyay both think 3D-printing technology will have a place in medicine.
At the Materials Research Group, Dr. Bose, Dr. Bandyopadhyay, and their graduate students run an impressive, interdisciplinary program. Dr. Bandyopadhyay stated that their work is “primarily an engineering-based solution to a biological problem.” The work, according to Dr. Bose, combines chemistry, materials characterization, materials processing and biological characterization.
When asked if we might see 3D printers in hospitals, ready to make custom grafts and prostheses, Dr. Bandyopadhyay says simply that “Co-location is not required.” He tells a story of working with CAD files generated from an MRI that he received from a neurosurgeon in India. Dr. Bandyopadhyay had never met the surgeon or the patient, but using the CAD files, he was able to print a custom titanium plate for a large section of the patient’s skull that was destroyed in a motorcycle crash.
“It takes one to two days to print a piece like that, and it takes up to one to two days to stabilize a patient after major trauma,” said Dr. Bandyopadhyay.
“Use of 3D printing can be used in a much broader sense, we can also develop a remedy or structure for a defect-specific or patient-specific bone defect. If the doctor provides a scan, we can convert it to a CAD file and make the bone scaffold specific to the defect size or shape,” said Dr. Bose. With current bone graft and prosthesis manufacturing methods, making a device from a patient specific image or out of an unusual material, due to a patient allergy to chromium for example, is prohibitively expensive and time consuming. Dr. Bose and Dr. Bandyopadhyay are working on making 3D printers into a cheaper, faster and more precise manufacturing methods.
The Group also works with WSU professors of veterinary medicine for testing the viability of their grafts and prostheses. Graduate students work on all aspects of the research, from imaging to CAD to 3D printing to veterinary surgery, explains Dr. Bandyopadhyay.

A walk through the Materials Research Group’s labs reveals the sequence of the process for printing and testing the ceramic and metal. The first room holds several 3D printers, one for ceramic powder and polymer binder, one for metal powder and one for just polymer. The next room houses a plasma sprayer for further refining products with additional coatings. And finally, a room for cell culture, where cells are grown for introduction to printed objects. Seeing where each of these phases happens gives an appreciation of how much Dr. Bose and Dr. Bandyopadhyay have accomplished since 1998.
But what about using 3D printers to build living things, with cell-based inks? A number of researchers at universities and private companies are doing just that.
 Looking to revolutionize how doctors obtain organs and tissues, specialists in regenerative medicine are using 3D printers with cell-based ink in conjunction with existing tissue culture techniques to create organs and tissues that could save, extend, and improve patients’ lives. 3D printers with ink containing cultured cells deposit at least one type of cell on a protein scaffold or within a mold made of a dissolvable gel. When the printed tissues are kept in an environment with temperature and oxygen levels resembling those inside a living animal, the natural embryonic development processes make cells grow together, just as happens in living animals.
Looking to revolutionize how doctors obtain organs and tissues, specialists in regenerative medicine are using 3D printers with cell-based ink in conjunction with existing tissue culture techniques to create organs and tissues that could save, extend, and improve patients’ lives. 3D printers with ink containing cultured cells deposit at least one type of cell on a protein scaffold or within a mold made of a dissolvable gel. When the printed tissues are kept in an environment with temperature and oxygen levels resembling those inside a living animal, the natural embryonic development processes make cells grow together, just as happens in living animals.
Professor Gabor Forgacs, of the University of Missouri-Columbia’s Biological Physics department, founded San Diego-based company Organovo in 2007. Organovo began operations in 2009, seeking to use 3D-printing technology in regenerative medicine.
Organovo uses 3D printers to assemble 2D layers of cellular material corralled within a mold of non-invasive gel. Cellular self-assembly takes over and leads to the 2D layers flowing together, leaving the tissue with the desired shape and function. The non-invasive gel allows Organovo to print and grow tissue without using a protein scaffold for printed cells to adhere to and grow on. Protein scaffolds, whether made from an existing organ stripped of its cells or created in a lab, have been a staple of other efforts to cultivate organs and tissues.
On April 22, 2013, Organovo announced that it had created the world’s “first fully cellular 3D bioprinted liver tissue.” This multicellular material is able to perform many functions of a natural liver, including cholesterol biosynthesis, enzymatic activities, and production of other compounds. Organovo stated the liver tissue is up to 500 microns and 20 cells thick, and exhibits “tissue-like cellular density.”
Organovo has developed its own 3D printer and software package for scaffold-free bioprinting, the NovoGen MMX Bioprinter. While Organovo has yet to produce any tissue that has gone into a patient, pharmaceutical researchers are already using the NovoGen MMX Bioprinter to create human tissues for drug development and disease modeling. In February 2013, Organovo and ZenBio announced a partnership for using ZenBio’s expertise in providing human cell solutions with Organovo’s printing technology. Organovo offers training and support to companies that purchase the printer. In December 2012, Organovo announced a collaboration with Autodesk “to create the first 3D design software for bioprinting.”
Besides Organovo, several universities have been developing 3D bioprinting technology. Chief among these are the Wake Forest Institute for Regenerative Medicine (WFIRM), Cornell University and the University of Iowa.
 WFIRM was the first facility in the world to transplant a lab-grown organ into a human. Dr. Anthony Atala, chair and director of the institute as well as the head of the Wake Forest Urology Department, has transplanted “hundreds” of lab-grown bladders into patients since the early 2000s. WFIRM does not use a printer for these bladders; rather they use a biopsy from the patient’s bladder to culture cells around a scaffold, and then incubate the bladder in a chamber whose temperature and oxygen level are meant to mimic conditions inside a healthy human.
WFIRM was the first facility in the world to transplant a lab-grown organ into a human. Dr. Anthony Atala, chair and director of the institute as well as the head of the Wake Forest Urology Department, has transplanted “hundreds” of lab-grown bladders into patients since the early 2000s. WFIRM does not use a printer for these bladders; rather they use a biopsy from the patient’s bladder to culture cells around a scaffold, and then incubate the bladder in a chamber whose temperature and oxygen level are meant to mimic conditions inside a healthy human.
WFIRM has now pioneered use of printers in its regenerative medicine labs. Researchers have used a modified inkjet printer, with solution of cells in the ink cartridge, to print organs, including a mouse heart. WFIRM researchers have developed techniques to grow large areas of skin from human foreskins, and are now developing a vial-based scan-and-print system to apply skin cells to burns that would normally require taking skin grafts from other areas on a patient’s body. In lab experiments, wounds on mice treated with printed skin cells healed in three weeks, versus five weeks for mice not treated with the printed skin.
Now, WFIRM is combining its expertise in culturing cells and using inkjet printers with cell-based inks to use 3D printers to create major organs with multiple tissue types. The 3D printer uses morphometric models based on CT scans. Dr. Atala brought one of WFIRM’s customized 3D printers to a 2011 TED Talk and presented a human kidney that was printed backstage. While the organ Dr. Atala presented in his talk is not suitable for transplant, it shows the amazing potential of 3D printers to create organs for transplants that do not require a finding and transporting an organ from a donor. Since a 3D printer can use ink created from a patient’s own cells, donor compatibility issues and threats to transplanted organs from the patient’s immune system will disappear.
The University of Iowa’s Biomanufacturing Laboratory, part of the Mechanical and Industrial Department, has Dr. Ibrahim Ozbolat using the world’s only multi-arm 3D bio-printer to make organs. The multi-arm printer, developed at the University of Iowa’s Center for Computer Aided Design’s Advanced Manufacturing Technology group, or AMTech, can print complex structures using different kinds of cells simultaneously. For example, one arm can print vascular tissue while another arm prints tissue specific to the organ being printed, such as the glucose-sensitive pancreatic tissue.
Ozbolat is a co-director at AMTech, and he hopes to print usable organs within five to ten years. AMTech is currently focusing on creating pancreatic tissue, using an interdisciplinary team that includes specialists in medicine, biology, anatomy, mechanical engineering, robotics and computer aided design. According to Ozbolat, creating blood vessels is essential to printing any tissue that is more than one centimeter thick, to keep cells from dying.
At Cornell University, Dr. Lawrence Bonassar of the Biomedical Engineering Department recently made news when his lab used a 3D printer to create a human earlobe. The process begins by scanning the patient’s head in order to create a computer model of the ear’s shape. This model then directs a 3D printer using a cell-based ink to print the ear. Ears are a good option for printing, because cartilage does not require a blood supply.

Stepping away from medical uses for 3D bioprinting, Professor Gabor Forgacs of Organovo and his son Andras founded Modern Meadow with Dr. Francoise Marga and Dr. Karoly Jakab in 2011. The Columbia, Mo.-based company wants to use 3D printers and other processes that allow for large-scale production to make meat and leather. Modern Meadow’s founders believe that meat and leather printed from lab-grown cells can provide safer, more environmentally friendly animal products than farming and ranching.
Modern Meadow’s process starts with a biopsy from a healthy animal, taking skin for leather, or muscle, fat and other tissues for meat. Modern Meadow claims that cultured meat requires 99 percent less land, 96 percent less water and 45 percent less energy than meat from livestock. In addition, cultured meat produces 96 percent less greenhouse gas emissions, and does not risk spreading livestock-borne disease.
“If we can get this technology right, and if we can mainstream it, then it supports a different, positive vision from science fiction, one where we can locally grow on-demand, cruelty free, sustainably abundant amounts of meat and leather in a way that can protect and preserve our future on this planet and beyond.”
Modern Meadow has made some meat and leather in their labs, but the products are far from ready for the marketplace. Modern Meadow’s plan is to test-market higher-end fashion products and gourmet meat before broadening into lower-priced markets as Modern Meadow’s production costs decrease and its reputation grows. Modern Meadow expects leather products to be ready before meat. Aside from the regulatory, cultural and engineering challenges to selling engineered meat, Gabor Forgacs explains in this TED Talk, where he eats a bite-sized morsel of printed pork, that Modern Meadow is still fine-tuning the fat content and electrical stimulation of muscle tissue during development that are necessary to make lab-grown meat taste like farm-raised meat.
This is just the beginning. Andras Forgacs explained in a Reddit IAMA, “Real steak is a big stretch. It won’t be the first product since steak is very hard to make for now. Instead, the first wave of meat products to be made with this approach will likely be minced meats (burgers, sausages, etc.) and pates (goose liver pate, etc.). Also seafood is an early possibility since the texture requires may be easier to achieve than premium cuts.”
Andras Forgacs closes a 2013 Solve for X presentation by summing up Modern Meadow’s goal nicely. “If we can get this technology right, and if we can mainstream it, then it supports a different, positive vision from science fiction, one where we can locally grow on-demand, cruelty free, sustainably, abundant amounts of meat and leather in a way that can protect and preserve our future on this planet and beyond.”
You won’t be eating a steak that comes from a 3D printer in 2013 or 2014, nor will you have a kidney, pancreas or bone graft from a 3D printer. But a number of academics and entrepreneurs are working on promising projects with 3D bioprinting that may well revolutionize our food industry and organ-transplant system within the next decade. It’s just a matter of time that some of us will be listening to the music of the spheres with our strange and revolutionary 3D-printed ears.
[Image: AFP Photo/Cornell University/Lindsay France]